Cell & Gene Therapy
Contract Development and Manufacturing Organisation (CDMO)
Product Development, Launch & Supply
Consulting Services
Enabling healthcare companies to launch and supply products
Cell & Gene Therapy | Pharmaceutical | Biotechnology | Medical Device
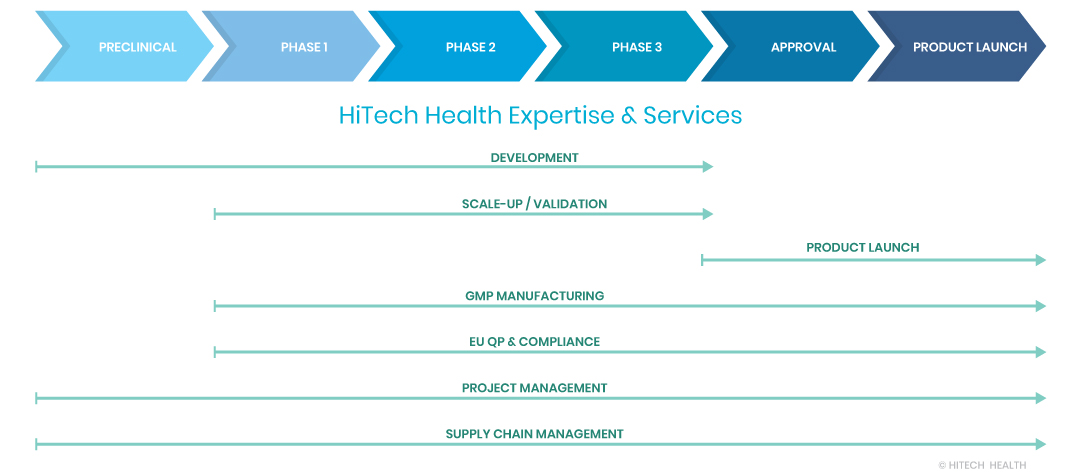
Your Commercialisation Journey
Our Clients
HiTech Health, University of Galway and Odyssey Validation Consultants awarded DTIF funding
The Disruptive Technologies Innovation Fund (DTIF) is a €500 million fund established under Ireland’s National Development Plan (NDP) in 2018. On Thursday 23rd November, Ireland’s Minister for Enterprise, Trade and Employment, Simon Coveney TD, Minister for Further...
Empowering Tomorrow’s Therapies: Hitech Health at Advanced Therapies Integrates 2023
Hitech Health is delighted to be attending Advanced Therapies Integrates in Stevenage, England on the 30th November. This event aims to drive the translation of cutting-edge research and disruptive innovation into the wide-spread adoption of Advanced Therapies,...
Afortiori Development and HiTech Health Forge an Important Collaboration
Afortiori Development and HiTech Health Forge an Important Collaboration to Support the Clinical Research and Product Development of Groundbreaking Cell and Gene Therapies. Galway, November 9, 2023 - Afortiori Development, a premier full-service Clinical Research...
Telephone
IE: +353-1-9631489
UK: +44-20-30267419
US: +1-857-3265835
Contact us
Get in touch anytime – we are always available to discuss your upcoming projects.











